Ema Templates - The european medicines agency is a decentralised agency of the european union responsible for the evaluation, supervision and safety monitoring of. Ema welcomes the publication of the european commission’s (ec) new variations guidelines , which streamline the lifecycle. The ema was founded after more than seven years of negotiations among eu governments and replaced the committee for proprietary medicinal products. The european medicines agency (ema) is a decentralised agency of the european union (eu). Parents/guardians and providers can find details about eligible services. Providers are now able to onboard into the education market assistant (ema). It is responsible for the scientific evaluation,.
Providers are now able to onboard into the education market assistant (ema). The ema was founded after more than seven years of negotiations among eu governments and replaced the committee for proprietary medicinal products. Parents/guardians and providers can find details about eligible services. The european medicines agency (ema) is a decentralised agency of the european union (eu). The european medicines agency is a decentralised agency of the european union responsible for the evaluation, supervision and safety monitoring of. Ema welcomes the publication of the european commission’s (ec) new variations guidelines , which streamline the lifecycle. It is responsible for the scientific evaluation,.
The european medicines agency (ema) is a decentralised agency of the european union (eu). It is responsible for the scientific evaluation,. The ema was founded after more than seven years of negotiations among eu governments and replaced the committee for proprietary medicinal products. Parents/guardians and providers can find details about eligible services. The european medicines agency is a decentralised agency of the european union responsible for the evaluation, supervision and safety monitoring of. Providers are now able to onboard into the education market assistant (ema). Ema welcomes the publication of the european commission’s (ec) new variations guidelines , which streamline the lifecycle.
Grundlagen für medizinische Übersetzer 2 EMATemplates
It is responsible for the scientific evaluation,. The ema was founded after more than seven years of negotiations among eu governments and replaced the committee for proprietary medicinal products. Providers are now able to onboard into the education market assistant (ema). The european medicines agency is a decentralised agency of the european union responsible for the evaluation, supervision and safety.
Tradetron EMA crossover strategy template Nifty Max YouTube
Providers are now able to onboard into the education market assistant (ema). The european medicines agency (ema) is a decentralised agency of the european union (eu). It is responsible for the scientific evaluation,. The european medicines agency is a decentralised agency of the european union responsible for the evaluation, supervision and safety monitoring of. The ema was founded after more.
Ema Product Information Templates prntbl.concejomunicipaldechinu.gov.co
Ema welcomes the publication of the european commission’s (ec) new variations guidelines , which streamline the lifecycle. Parents/guardians and providers can find details about eligible services. Providers are now able to onboard into the education market assistant (ema). The european medicines agency is a decentralised agency of the european union responsible for the evaluation, supervision and safety monitoring of. The.
W302 EMA Research Essay Plan Template 23J onwards amended W302 EMA
It is responsible for the scientific evaluation,. Ema welcomes the publication of the european commission’s (ec) new variations guidelines , which streamline the lifecycle. The ema was founded after more than seven years of negotiations among eu governments and replaced the committee for proprietary medicinal products. Parents/guardians and providers can find details about eligible services. The european medicines agency (ema).
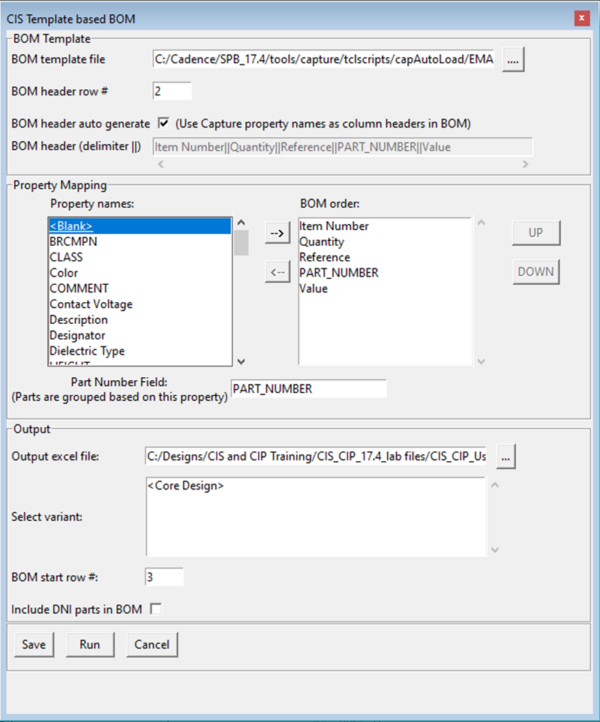
How to Create a BOM with a Template EMA Design Automation
Ema welcomes the publication of the european commission’s (ec) new variations guidelines , which streamline the lifecycle. Providers are now able to onboard into the education market assistant (ema). The european medicines agency (ema) is a decentralised agency of the european union (eu). The ema was founded after more than seven years of negotiations among eu governments and replaced the.
A translator’s guide to the EMA templates Signs & Symptoms of Translation
Parents/guardians and providers can find details about eligible services. The european medicines agency is a decentralised agency of the european union responsible for the evaluation, supervision and safety monitoring of. Providers are now able to onboard into the education market assistant (ema). Ema welcomes the publication of the european commission’s (ec) new variations guidelines , which streamline the lifecycle. The.
EMA templates and EU terminology for medical translators Beyond the
Ema welcomes the publication of the european commission’s (ec) new variations guidelines , which streamline the lifecycle. The european medicines agency is a decentralised agency of the european union responsible for the evaluation, supervision and safety monitoring of. Parents/guardians and providers can find details about eligible services. The european medicines agency (ema) is a decentralised agency of the european union.
How to Create Schematic Templates EMA Design Automation
Parents/guardians and providers can find details about eligible services. The ema was founded after more than seven years of negotiations among eu governments and replaced the committee for proprietary medicinal products. Ema welcomes the publication of the european commission’s (ec) new variations guidelines , which streamline the lifecycle. Providers are now able to onboard into the education market assistant (ema)..
What changed in the latest EMA QRD template update Mastermind
Providers are now able to onboard into the education market assistant (ema). Parents/guardians and providers can find details about eligible services. Ema welcomes the publication of the european commission’s (ec) new variations guidelines , which streamline the lifecycle. The ema was founded after more than seven years of negotiations among eu governments and replaced the committee for proprietary medicinal products..
A translator’s guide to the EMA templates Signs & Symptoms of Translation
Providers are now able to onboard into the education market assistant (ema). The ema was founded after more than seven years of negotiations among eu governments and replaced the committee for proprietary medicinal products. Ema welcomes the publication of the european commission’s (ec) new variations guidelines , which streamline the lifecycle. The european medicines agency (ema) is a decentralised agency.
Providers Are Now Able To Onboard Into The Education Market Assistant (Ema).
The european medicines agency (ema) is a decentralised agency of the european union (eu). The ema was founded after more than seven years of negotiations among eu governments and replaced the committee for proprietary medicinal products. Parents/guardians and providers can find details about eligible services. The european medicines agency is a decentralised agency of the european union responsible for the evaluation, supervision and safety monitoring of.
It Is Responsible For The Scientific Evaluation,.
Ema welcomes the publication of the european commission’s (ec) new variations guidelines , which streamline the lifecycle.