Stoichiometry Sheet 6 Answers - According to this law, the total mass of the reactants is equal to that of the. The stoichiometry of a reaction describes the relative amounts of reactants and products in a balanced chemical equation. Stoichiometry (/ ˌstɔɪkiˈɒmɪtri / ⓘ) is the relationships between the quantities of reactants and products before, during, and following. It also shows the numerical relationships between the reactants. Stoichiometry is like the recipe for a cake, but instead of baking, we’re dealing with chemical reactions. It shows what reactants (the ingredients) combine to form what products (the cookies). Stoichiometry is based on the principle of the law of conservation of mass.
It also shows the numerical relationships between the reactants. Stoichiometry (/ ˌstɔɪkiˈɒmɪtri / ⓘ) is the relationships between the quantities of reactants and products before, during, and following. Stoichiometry is like the recipe for a cake, but instead of baking, we’re dealing with chemical reactions. Stoichiometry is based on the principle of the law of conservation of mass. It shows what reactants (the ingredients) combine to form what products (the cookies). The stoichiometry of a reaction describes the relative amounts of reactants and products in a balanced chemical equation. According to this law, the total mass of the reactants is equal to that of the.
The stoichiometry of a reaction describes the relative amounts of reactants and products in a balanced chemical equation. According to this law, the total mass of the reactants is equal to that of the. It also shows the numerical relationships between the reactants. Stoichiometry is like the recipe for a cake, but instead of baking, we’re dealing with chemical reactions. Stoichiometry is based on the principle of the law of conservation of mass. It shows what reactants (the ingredients) combine to form what products (the cookies). Stoichiometry (/ ˌstɔɪkiˈɒmɪtri / ⓘ) is the relationships between the quantities of reactants and products before, during, and following.
SOLUTION Chemistry stoichiometry practice problems with answers
According to this law, the total mass of the reactants is equal to that of the. It shows what reactants (the ingredients) combine to form what products (the cookies). Stoichiometry (/ ˌstɔɪkiˈɒmɪtri / ⓘ) is the relationships between the quantities of reactants and products before, during, and following. It also shows the numerical relationships between the reactants. Stoichiometry is based.
Practice stoichiometry PDF Worksheets Library
It also shows the numerical relationships between the reactants. The stoichiometry of a reaction describes the relative amounts of reactants and products in a balanced chemical equation. Stoichiometry is like the recipe for a cake, but instead of baking, we’re dealing with chemical reactions. Stoichiometry (/ ˌstɔɪkiˈɒmɪtri / ⓘ) is the relationships between the quantities of reactants and products before,.
Free Printable Stoichiometry Worksheets, 60 OFF
It shows what reactants (the ingredients) combine to form what products (the cookies). According to this law, the total mass of the reactants is equal to that of the. The stoichiometry of a reaction describes the relative amounts of reactants and products in a balanced chemical equation. Stoichiometry is like the recipe for a cake, but instead of baking, we’re.
Stoichiometry Cheat Sheet Exams Stoichiometry Docsity
Stoichiometry is based on the principle of the law of conservation of mass. It also shows the numerical relationships between the reactants. Stoichiometry (/ ˌstɔɪkiˈɒmɪtri / ⓘ) is the relationships between the quantities of reactants and products before, during, and following. Stoichiometry is like the recipe for a cake, but instead of baking, we’re dealing with chemical reactions. According to.
Copy w answers Stoichiometry unit study guide Stoichiometry Unit
It shows what reactants (the ingredients) combine to form what products (the cookies). According to this law, the total mass of the reactants is equal to that of the. Stoichiometry (/ ˌstɔɪkiˈɒmɪtri / ⓘ) is the relationships between the quantities of reactants and products before, during, and following. It also shows the numerical relationships between the reactants. The stoichiometry of.
Stoichiometry Practice 6 problems ANSWERS PDF
According to this law, the total mass of the reactants is equal to that of the. It also shows the numerical relationships between the reactants. Stoichiometry is like the recipe for a cake, but instead of baking, we’re dealing with chemical reactions. Stoichiometry (/ ˌstɔɪkiˈɒmɪtri / ⓘ) is the relationships between the quantities of reactants and products before, during, and.
Percent Yield Stoichiometry Chemistry Worksheet 10 Problems with
It also shows the numerical relationships between the reactants. Stoichiometry is like the recipe for a cake, but instead of baking, we’re dealing with chemical reactions. Stoichiometry is based on the principle of the law of conservation of mass. According to this law, the total mass of the reactants is equal to that of the. Stoichiometry (/ ˌstɔɪkiˈɒmɪtri / ⓘ).
Key Worksheet Chemical Reactions And Stoichiometry With
Stoichiometry is like the recipe for a cake, but instead of baking, we’re dealing with chemical reactions. Stoichiometry (/ ˌstɔɪkiˈɒmɪtri / ⓘ) is the relationships between the quantities of reactants and products before, during, and following. It also shows the numerical relationships between the reactants. Stoichiometry is based on the principle of the law of conservation of mass. The stoichiometry.
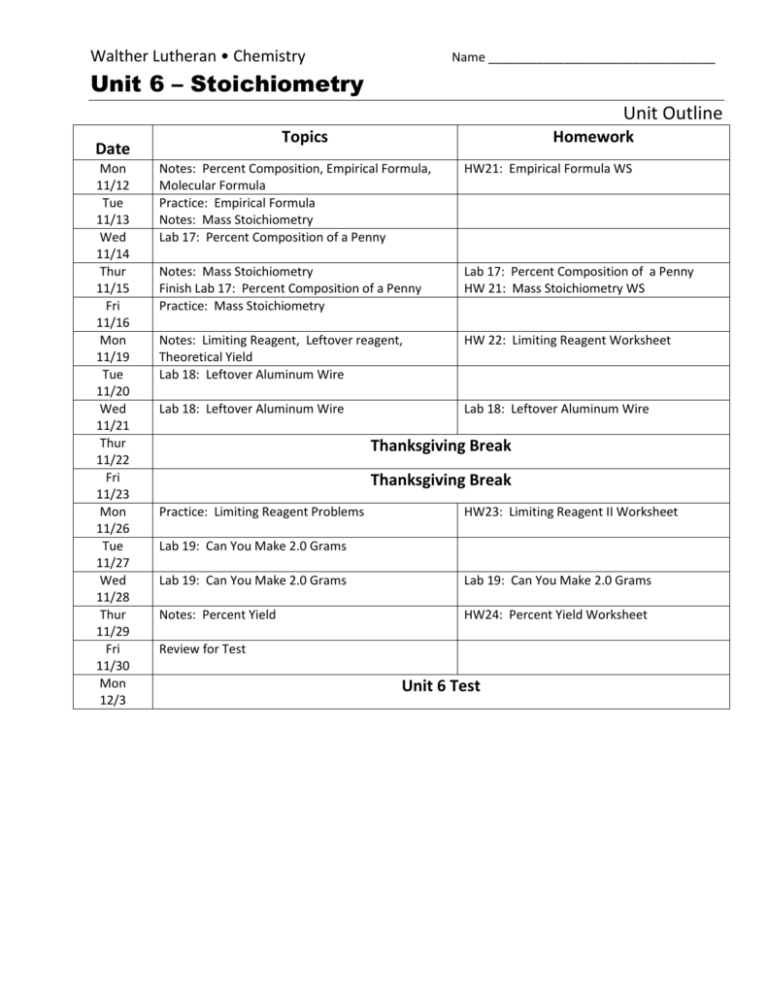
Stoichiometry Unit Outline High School Chemistry
The stoichiometry of a reaction describes the relative amounts of reactants and products in a balanced chemical equation. It also shows the numerical relationships between the reactants. Stoichiometry (/ ˌstɔɪkiˈɒmɪtri / ⓘ) is the relationships between the quantities of reactants and products before, during, and following. It shows what reactants (the ingredients) combine to form what products (the cookies). According.
Stoichiometry Worksheet 2 Answers
Stoichiometry is based on the principle of the law of conservation of mass. The stoichiometry of a reaction describes the relative amounts of reactants and products in a balanced chemical equation. It also shows the numerical relationships between the reactants. It shows what reactants (the ingredients) combine to form what products (the cookies). Stoichiometry (/ ˌstɔɪkiˈɒmɪtri / ⓘ) is the.
It Also Shows The Numerical Relationships Between The Reactants.
Stoichiometry is like the recipe for a cake, but instead of baking, we’re dealing with chemical reactions. Stoichiometry is based on the principle of the law of conservation of mass. According to this law, the total mass of the reactants is equal to that of the. Stoichiometry (/ ˌstɔɪkiˈɒmɪtri / ⓘ) is the relationships between the quantities of reactants and products before, during, and following.
The Stoichiometry Of A Reaction Describes The Relative Amounts Of Reactants And Products In A Balanced Chemical Equation.
It shows what reactants (the ingredients) combine to form what products (the cookies).